ABSTRACT
Mechanical cell stimulation has only recently been highlighted for its ability to direct cell function and fate. The development of techniques to apply controlled mechanical stimulation to cells has allowed researchers to examine the effects of forces on cell behavior. These studies have shown that cells, and in particular, stem cells are very sensitive to mechanical stimuli.
While the potential impact of mechanical forces has been demonstrated by these studies, there have not been many studies that compare the differing effects of stimulation profiles and met hods. Further, molecular mechano sensing mechanisms have not been identified. In this work, we compared the effects of cyclic tensile stretch and fluid flow induced shear on mesenchymal stem cell (MSC) adipogenesis. We also implicated extra cellular signal-regulated kinase (ERK) phosphorylation as a molecular mechanism involved in both cases.
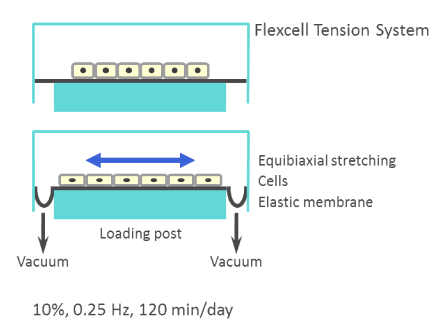
To test mechanical control of MSC adipogenesis, C3H10T1/2 murine MSCs were exposed to cyclic equibiaxial tensile stretch at 10% strain or 20 dyne/cm2 of fluid flow induced shear and induced to differentiate. For this, Flexcell FX-5000 cell stretching device and Flexcell Streamer fluid flow devices were used. Interestingly, lipid accumulation, a marker of MSC adipogenesis, was suppressed by both mechanical stretch and flow shear. Also, genetic markers for MSC adipogenic commitment (PPARγ, aP2, and C/EBPα) were down regulated by cell stretch.
We observed as a potential mechanotransduction mechanism that both stimulation methods induced significant ERK1/2 phosphorylation in MSCs. Furthermore, both stretch and flow induced mechanical suppression of MSC adipogenesis was deteriorated in the presence of PD98059, an established pharmacological ERK inhibitor.
Combined, these results indicate that cell stretch and fluid flow suppress adipogenic commitment and differentiation of MSCs, potentially via the mechanical activation of ERK signaling. While more comparisons of varying regimens are required, our data suggests that both cell stretch and fluid flow have potential to down regulate MSC adipogenesis and may share a similar cell mechano transduction mechanism such as ERK. Further, inhibiting MSC adipogenesis via mechanical cues may provide new insights for obesity research.
LITERATURE REVIEW
On a clinical level, obesity is characterized by a body mass index of greater than 30 in adults. As an individual reaches and exceeds that level, the incidence of heart disease, diabetes, liver steatosis, and several cancers rises dramatically (Cornier, et al., 2008; Shoham and Gefen, 2012). On a population scale, the most effective measures have focused on diet and exercise. However the complex interactions of the cellular processes that control the risk for and development of obesity are not well understood.
Mechanical Control:
Recently there has been increased interest in mechanophysical control of cell behavior. Studies have been devised to probe the effect of substrate topography, cell micronfinement, forced cell alignment, mechanical stretch, fluid flow, and hydrostatic pressure on cell behavior. The mechanophysical signals do not always produce similar cell outcomes. For example, it was demonstrated that cyclic pressure and fluid flow differentially affect the actin network of osteoblasts, causing vastly different responses in subsequent signaling events (Gardinier, et al., 2009).
Fluid flow:
Fluid flow has been proposed as a potential regulator of cell function for many years. Early efforts focused on biomimetics in an attempt to develop flow bioreactors for regenerative medicine purposes and in particular functional vascular tissue (Radisic, et al., 2007). With recent advances in engineering tools including finite elemental analysis, one can more accurately analyze the minute differences in forces and perform systematic studies. This allows us to control more of the variables and isolate the effect of differing levels and profiles of shear.
STRETCH
Experimental Design:
The stretch portion of this experiment follows a design describe below. First we test whether stretch decreases MSC adipogenic commitment induced by BMP4 via assessing Oil Red O lipid staining and gene expression. Next we examine if stretch increases ERK phosphorylation and compare with BMP4 treatment alone case. Finally we test the effect of blocking ERK activation by PD98059 on the stretch induced MSC adipogenesis inhibition.
Mechanical Stimulation:
Cells were subjected to stretch during the BMP4 pretreatment period to examine the mechanical control of BMP4 induction. The FX-5000 tension system (Flexcell) was used to stretch the collagen coated silicon membrane on which cells are cultured. A computer-controlled system of valves allowed for precise control of vacuum pressure on the membrane to achieve required membrane deformation (and thus required strain).
FLUID FLOW
Mechanical Stimulation:
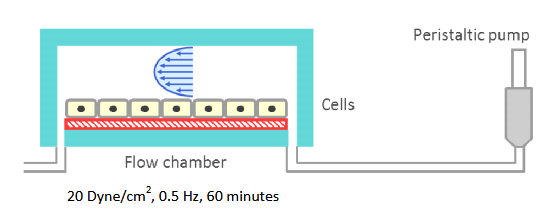
Experimental groups were subjected to flow shear on day 0 using the Flexcell Streamer system (Flexcell International). The schematic is showapplied to the cells 20 dyne/cm 2 of fluid shear oscillating at 0.5 Hz for 60 minutes.
This system uses a peristaltic pump to drive culture media across the cells at a constant volumetric flow rate. Upstream to the flow chamber are a series of bubble traps and the OsciFlow device (Flexcell International). The bubble traps dramatically decrease delamination and prevent the destruction of the samples due to unstable flow. The OsciFlow device will be explained in the next paragraph.
Data Collection:
To assess adipogenic differentiation, Oil Red O staining was used. As carried out in chapter two for stretched cells, Oil red O stained cells were imaged and the amount of synthesized lipids were quantified by a spectrophotometer. All the lipid staining data points were normalized to untreated static control.
CONCLUSION
In conclusion, we showed that mechanical stimulation plays a pivotal role in the commitment and differentiation of mesenchymal stem cells. Specifically, both stretch and fluid flow stimulations showed a strong potential to decrease MSC adipogenesis. This was demonstrated by (1) cell stretch inhibition of BMP4 modulated up regulation of adipogenic genes and lipid synthesis and (2) fluid flow suppression of PPARγ dependent lipid synthesis, both for C3H10T1/2 MSCs.
Importantly, our data demonstrated that ERK1/2 phosphorylation may be significantly involved in both cases, as demonstrated by the observations that under ERK inhibitor mechanical inhibition of MSC adipogenesis was disrupted for both stretch and fluid flow stimulations.
Future research could probe more systematically the mechanical stimulation regimens, for both stretch and fluid flow. Also, fluid flow inhibition of BMP4 induced adipogenic commitment may also be tested. One may also attempt to isolate the effect of membrane and cell deformation from the shear force provided by the media under cell stretch.
We have demonstrated the complex nature of MSC adipogenic commitment, which is dependent on both soluble and mechanical environments, and proposed a potential regulatory mechanism, e.g., ERK, to inhibit MSC adipogenesis and thus to target in the ongoing fight against obesity and obesity related diseases.
Source: University of Nebraska-Lincoln
Author: Daniel E. Menter